November 20, 2022
Innovating for Tomorrow
In 2002, a small group of research scientists, chemists and biologists working in immunology formed a drug discovery and development team. They shared a vision of creating a biopharmaceutical company willing to tackle difficult medical problems, with the end goal of making a significant impact for patients.
Twenty years later, this focus remains unchanged. For Incyte, it has, and always will be, about following the science. If we believe we have a meaningful solution for a medical need, we will pursue it—even if it is for a rare indication.
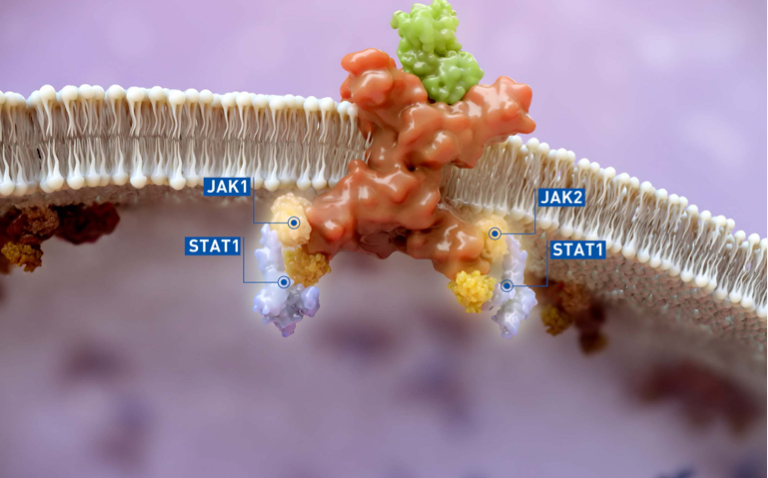
Building on our heritage and deep understanding of the impact of the immune system on disease, the result of these efforts has been profound. We focus on developing medicines in areas we can provide benefit beyond the current standard of care, or areas of need with limited or no options. These efforts have resulted in multiple therapies for patients in Oncology and Inflammation & Autoimmunity, including several that have been the first approved medicines in their class or indication.
Looking ahead, we hope to expand our pipeline and advance our key clinical programs in areas of significant unmet need. We are progressing a number of Phase 2 and Phase 3 clinical trials in a program called LIMBER. LIMBER was designed to evaluate ruxolitinib combination strategies—as well as monotherapy strategies for other compounds—for patients with myeloproliferative neoplasms (MPNs), a group of rare, chronic and progressive blood cancers, and graft-versus-host disease (GVHD). Additionally, in 2022 we have multiple key development programs progressing and anticipate important new data from our pipeline. This year we also received approval in the U.S. of the first and only FDA-approved product for repigmentation in nonsegmental vitiligo, a chronic autoimmune condition that affects millions of people and is characterized by depigmentation of skin. We expect a regulatory decision in Europe for this therapy sometime next year.
Reflecting the growth of our portfolio, our team has grown over the last 20 years to capitalize on these opportunities. Today, we are 2,200 employees strong, with a growing footprint at our headquarters in Wilmington, Delaware, and established offices across North America, Europe and Asia. We have expanded our laboratories and manufacturing facilities, ensuring we have access to the most state-of-the-art tools and technology as we work to identify innovative new therapies for patients.
We are excited to share more about who we are as a company and our drive to help patient communities in new and different ways. We invite you to visit Solve On. Stories to join us on the journey as we continue to Solve On.
We can’t wait to see what the next 20 years of scientific excellence and innovation will bring.